Corona Covid-19 New Testing Kit From BOSCH Only 2.5 Hr Know the Result -Mr and Mrs Tamilan
The coronavirus SARS-CoV-2 is posing major challenges for healthcare systems and medical institutions worldwide. An ability to rapidly diagnose the virus is of invaluable help in curbing its exponential spread in many countries. Bosch’s new, fully automated rapid test for COVID-19 can help medical facilities such as doctors’ offices, hospitals, laboratories, and health centers make fast diagnoses.
The rapid molecular diagnostic test runs on the Vivalytic analysis device from Bosch Healthcare Solutions. “We want the Bosch rapid COVID-19 test to play a part in containing the coronavirus pandemic as quickly as possible. It will speed up the identification and isolation of infected patients,” says Dr. Volkmar Denner, chairman of the board of management of Robert Bosch GmbH.

Faster certainty, slower spread
Developed in just six weeks, the rapid test can detect a SARS-CoV-2 coronavirus infection in patients in under two and a half hours — measured from the time the sample is taken to the time the result arrives. Another advantage of the rapid test is that it can be performed directly at the point of care. This eliminates the need to transport samples, which takes up valuable time. It also means patients quickly gain certainty about their state of health, while allowing infected individuals to be identified and isolated immediately. With the tests currently in use, patients must usually wait one to two days for a result. “Time is of the essence in the fight against coronavirus. Reliable, rapid diagnosis directly on site with no back and forth — that is the great advantage of our solution, which we see as another example of technology that is ‘Invented for life,’” Denner says.
Bosch’s rapid test is one of the world’s first fully automated molecular diagnostic tests that can be used directly by all medical institutions. What’s more, it allows a single sample to be tested not just for COVID-19 but also for nine other respiratory diseases, including influenza A and B, simultaneously. “The special feature of the Bosch test is that it offers differential diagnosis, which saves doctors the additional time needed for further tests. It also provides them with a reliable diagnosis quickly so they can then begin suitable treatment faster,” says Marc Meier, president of Bosch Healthcare Solutions GmbH. The newly developed test will be available in Germany starting in April, with other markets in Europe and elsewhere to follow

Bosch’s rapid COVID-19 test is the result of collaboration between the company’s Bosch Healthcare Solutions subsidiary and the Northern Irish medical technology company Randox Laboratories Ltd. “Together with our partner Randox, we have succeeded in developing this innovative rapid test within a very short time frame, and we are now in a position to offer it to the market. The Bosch Vivalytic analysis device evaluates the test safely and reliably directly in the hospital, in the lab, or in the doctor’s office, guaranteeing the best possible protection for patients and medical staff,” Meier says. The company is currently examining how it can help doctors and nursing staff in medical facilities such as the Robert Bosch Hospital get tested promptly so they can be fit to work for as long as possible — with no risk of infecting others.
Easy application at the point of care
In various laboratory tests with SARS-CoV-2, the Bosch test delivered results with an accuracy of over 95 percent. The rapid test meets the quality standards of the World Health Organization (WHO). A sample is taken from the patient’s nose or throat using a swab. Then the cartridge, which already contains all the reagents required for the test, is inserted into the Vivalytic device for analysis. During the analysis, medical staff can devote themselves to other tasks, for example treating patients. The Vivalytic analyzer is designed to be so user-friendly that even medical personnel who have not been specially trained on it can reliably perform the test.
A Bosch Vivalytic analyzer can perform up to ten tests in the space of 24 hours. This means it takes just 100 devices to evaluate up to 1,000 tests per day. Given the dynamic spread of the coronavirus SARS-CoV-2, laboratories are already working beyond capacity. The Bosch Vivalytic will thus help to increase available testing capacities.
VRI multiplex test
Rapid test for COVID-19 (SARS-CoV-2)
The Vivalytic VRI test (Viral Respiratory Tract Infections) checks the patient’s sample for the SARS CoV-2 virus, which can lead to COVID-19, as well as other possible viral respiratory diseases. To prevent the continued rapid spread of COVID-19, it is necessary to identify infectious patients with and without symptoms. With the Vivalytic VRI test, physicians can quickly and efficiently distinguish between different infections with very similar symptoms, like influenza, and immediately begin the appropriate treatment. The test delivers a reliable result based on WHO guidelines in less than 2.5 hours at the point of care, i.e. in close proximity to where the patient’s sample was collected. This means a time-consuming logistics chain, in which the samples have to be transported, is no longer necessary.
Sample material:
Nasopharyngeal swab
Nasopharyngeal swab
Principle:
Integration of sample preparation (including process controls), DNA amplification, detection (μArray) and analysis on a single cartridge
Integration of sample preparation (including process controls), DNA amplification, detection (μArray) and analysis on a single cartridge
Purpose:
Nucleic acid based testing for direct identification of viral pathogens
Nucleic acid based testing for direct identification of viral pathogens
The Vivalytic Workflow
The Vivalytic platform is so easy to use that it can be handled in any clinic or medical practice without the need for laboratory personnel.
The sample is taken from the patient’s nose or throat using a swab and placed in the cartridge without any complex preparation.
The Vivalytic analyser then fully automatically processes the cartridge, which already contains all the reagents required for the test.
Since the system in each cartridge is isolated, the risk of infection during handling is minimized.
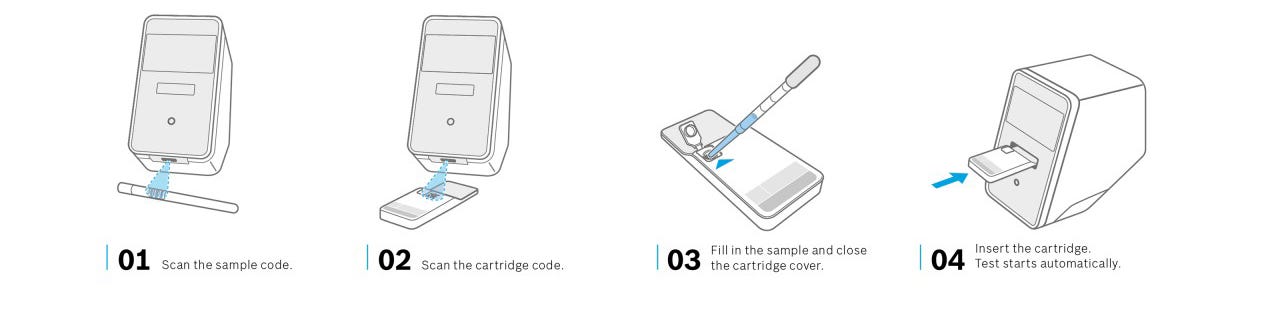
1.Collecting Samples

2.Taking Cartridge

3.Filling Sample into Cartridge

4.Run Test & Result

The Vivalytic analysers required for the COVID 19 rapid test can be purchased by medical institutions such as doctors’ surgeries, laboratories, hospitals and health centres. The test can be performed directly at the point of care.

The COVID-19-(SARS-CoV-2-) rapid testing kit for the Vivalytic platform developed by Bosch delivers reliable results at the point of care in less than 2.5 hours.